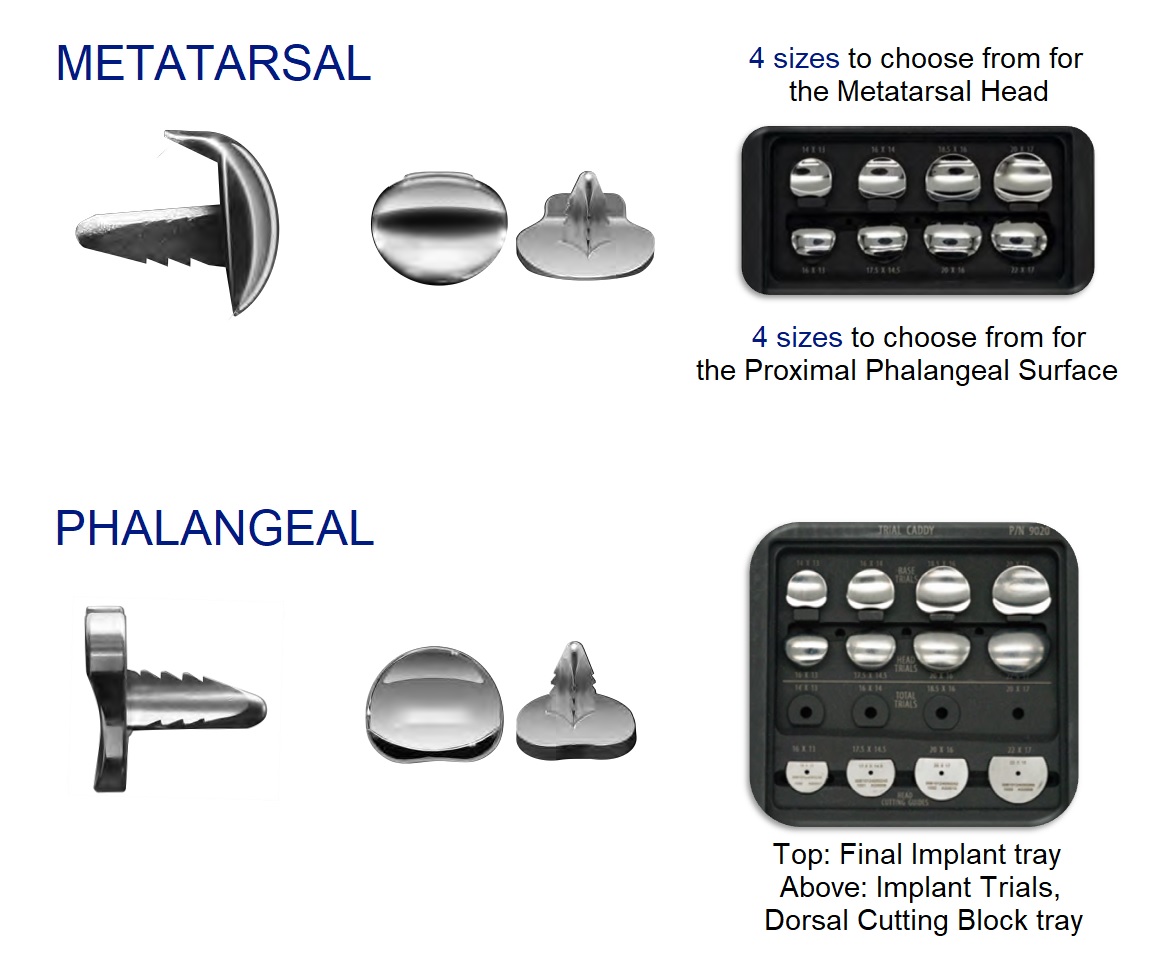
The Motion Toe System
FIRST METATARSAL PHALANGEAL JOINT • MINIMAL JOINT DISSECTION • MAXIMAL BONE PRESERVATION
The Motion Toe System is the most recently designed implant for the first metatarsal phalangeal joint. The design follows modern principles of joint replacement. Unlike many implants there is minimal joint dissection and maximal bone preservation. Many implants over dissect the joints leaving the ligament structures unbalanced.
Other implants remove too much bone causing early subsiding making the implant unstable and failure occurs more quickly. Moreover, if too much bone has been removed joint revision/salvage with fusion or another implant is more difficult. Most patients understand joint replacement failure occurs at all joints.
Because the Motion toe system is a resurfacing like many modern joint replacements the surgeon will more easily be able to revise the joint with another joint or fusion. Because the motion toe system is a modern design it more accurately replaces the joint surface. Many implants are designed with a round shape, the natural contour of the first metatarsal phalangeal joint is not round. The round shape may make the joint unstable after replacement. Having a natural shape allows the other side to articulate more naturally. A poorly sized implant on the head of the first metatarsal often will subside into the base of the proximal phalanx.
Some implants were designed for other parts of the body, this implant was designed solely for the first metatarsal phalangeal joint.
The Motion Toe System
State-of-the-art
•
Adheres to contemporary
principles of joint replacement
•
Prioritizes minimal joint
dissection and maximizes
bone preservation
•
Addressing common issues of
ligament imbalance and early
subsidence
MINIMAL JOINT DISSECTION
•
MAXIMAL BONE PRESERVATION

This publication sets forth detailed recommended procedures for using the Motion Toe System. It offers guidance that you should follow, but each surgeon must consider the particular needs of each patient and make appropriate adjustments as needed and as required. A workshop training is recommended prior to performing this surgery. All non-sterile devices must be cleaned and sterilize following your hospitals guidelines. Follow the instructions provided in our sterilization guide. Multi-component instruments must be properly disassembled for cleaning and reassembled as required. Please refer to the corresponding assembly/ disassembly instructions. The surgeon must discuss all relevant risks including the limited lifetime of the implant with the patient when necessary.
Indications: implant is designed to replace either side or the total of the first metatarsal phalangeal joint. The Hemi base with the poly is expected to be released 2024. For now, it will be a Hemi Head and a Hemi Base system. Replacement should occur at the surgeon’s discretion for arthritis of the first toe joint, except in instances of septic arthritis and disease states that greatly weaken the surrounding bone and tissues. Surgeon must be sure the patient is capable of undergoing the surgery and would benefit from the surgery. The implant is not to be reused and its use must follow FDA guidelines.
Contraindications:
• Acute or chronic infections, local or systemic
• Surgical procedures other than those mentioned
• Do not use on patients having known allergies these materials. If an allergy is suspected to a material in the product this must be ruled out prior to implantation
• Poor or insufficient bone stock or surrounding tissues, including skin and tissue coverage of the device once it is implanted
• The presence of any structural or functional abnormalities that would risk a positive outcome for the patient
• Other conditions that may place the patient at risk during or after the surgery
Precautions:
It is unknown what affect the implant will have on the patient’s normal function or on other issues the patient may have included prior joint replacement, proximal joint replacement, contralateral replacements in the other limb, use around MRI or CT scanners, etc. This first metatarsal phalangeal joint replacement system allows the surgeon to make decisions at the time of surgery for the benefit of the patient. Often x-rays are misleading and the surgical view shows the joint is worse than the surgeon anticipated or that one side of the joint is not as bad as the other. This is not intended to correct deformity in the joint in any plane, horizontal, vertical or frontal. If there is need for correction of the plane of deformity that needs to be done prior to application of the implant system. It is not intended to be placed in area of prior infection or other bone disease that can compromise the implant and its function.
Introducing the Motion Toe System, a state-of-the-art implant meticulously crafted for the first metatarsal phalangeal joint. This innovative design adheres to contemporary principles of joint replacement, setting it apart from conventional implants. It prioritizes minimal joint dissection and
maximizes bone preservation, addressing common issues of ligament imbalance and early subsidence seen in other implants.
Furthermore, the Motion Toe System’s unique resurfacing approach facilitates easier joint revision or fusion, while its anatomically accurate design ensures a stable and natural joint surface. Unlike round-shaped implants, which may compromise joint stability, this implant harmonizes with the joint’s natural contour, promoting smoother articulation. Designed exclusively for the first metatarsal phalangeal joint, it eliminates the risks
associated with implants meant for other anatomical regions.